Calculating formal charge co11/21/2023  
Bonding electrons are divided equally between the two bonded atoms, so one electron from each bond goes to each atom.Non-bonding electrons are assigned to the atom on which they are located.To calculate formal charges, we assign electrons in the molecule to individual atoms according to these rules: Formal charge is assigned to an atom in a molecule by assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. In this course, you will certainly need to be able to recognize whether a given species carries a charge (i.e., is an ion).Ī formal charge compares the number of electrons around a "neutral atom" (an atom not in a molecule) versus the number of electrons around an atom in a molecule. Too much emphasis can easily be placed on the concept of formal charge, and the mathematical approach is hard to justify. It is sometimes possible to write more than one Lewis structure for a substance that does not violate the octet rule, as we saw for CH 2O, but not every Lewis structure may be equally reasonable.\) Now let’s apply this procedure to some particular compounds, beginning with one we have already discussed. This will not change the number of electrons on the terminal atoms. If the central atom has fewer electrons than an octet, use lone pairs from terminal atoms to form multiple (double or triple) bonds to the central atom to achieve an octet.We will explain later that some atoms are able to accommodate more than eight electrons. If any electrons are left over, place them on the central atom.These electrons will usually be lone pairs. Beginning with the terminal atoms, add enough electrons to each atom to give each atom an octet (two for hydrogen).In H 2O, for example, there is a bonding pair of electrons between oxygen and each hydrogen. 
Place a bonding pair of electrons between each pair of adjacent atoms to give a single bond.For CO 3 2 −, for example, we add two electrons to the total because of the −2 charge. 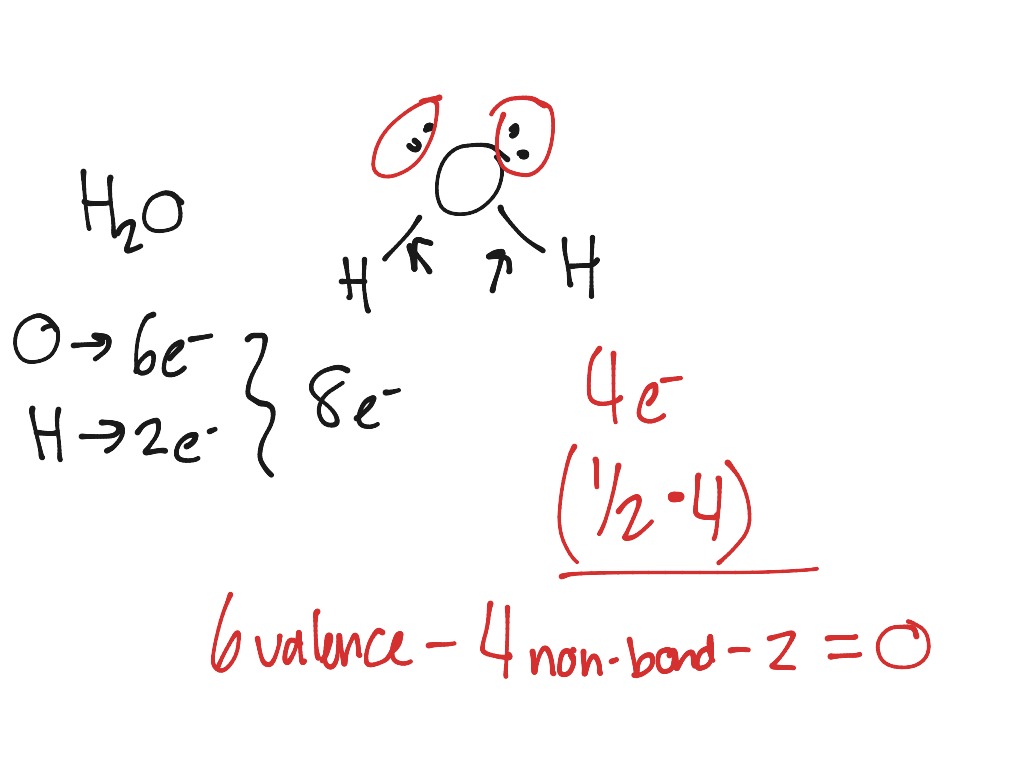
(Recall that the number of valence electrons is indicated by the position of the element in the periodic table.) If the species is a polyatomic ion, remember to add or subtract the number of electrons necessary to give the total charge on the ion. Add together the valence electrons from each atom.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
